Als new findings12/19/2023  
“We showed using multiple different approaches that the molecule we designed - the ‘bait’ -binds very highly and specifically to this poly-GR protein, and in binding to it prevents it from binding to ribosomal RNA and prevents it from going into the nucleus, which is where it becomes most toxic,” Kiskinis said. In both in vivo models and in iPSC neurons derived from patients with C9ORF72 mutations, the bait molecule inhibited toxicity. Using these findings, the investigators then designed an RNA “bait,” which tricks poly-GR into binding with something that appears to be ribosomal RNA -or in this case, the bait. Right: iPSC-derived motor neuron transduced with GFP-(GR)50 lentivirus, and stained for rRNA, showing the total overlap of poly-GR and rRNA in human motor neurons. Left: Neuron at the motor cortex of a C9ORF72 patient stained for MAP2 and rRNA, showing the pattern of expression of ribosomal RNA in a human neuron mostly at the cytosol and the nucleolus. Specifically, the poly-GR dipeptide binds to ribosomal RNA, which impairs ribosomal homeostasis, a process essential for cell differentiation and the overall makeup of a cell. Then, they used a technique called crosslinking immunoprecipitation, or CLIP-seq, to isolate specific RNA fragments and found that R-DPRs bind exclusively to ribosomal RNAs. 
Using computational and experimental techniques, the investigators found that R-DPRs have a strong binding affinity for RNA molecules. 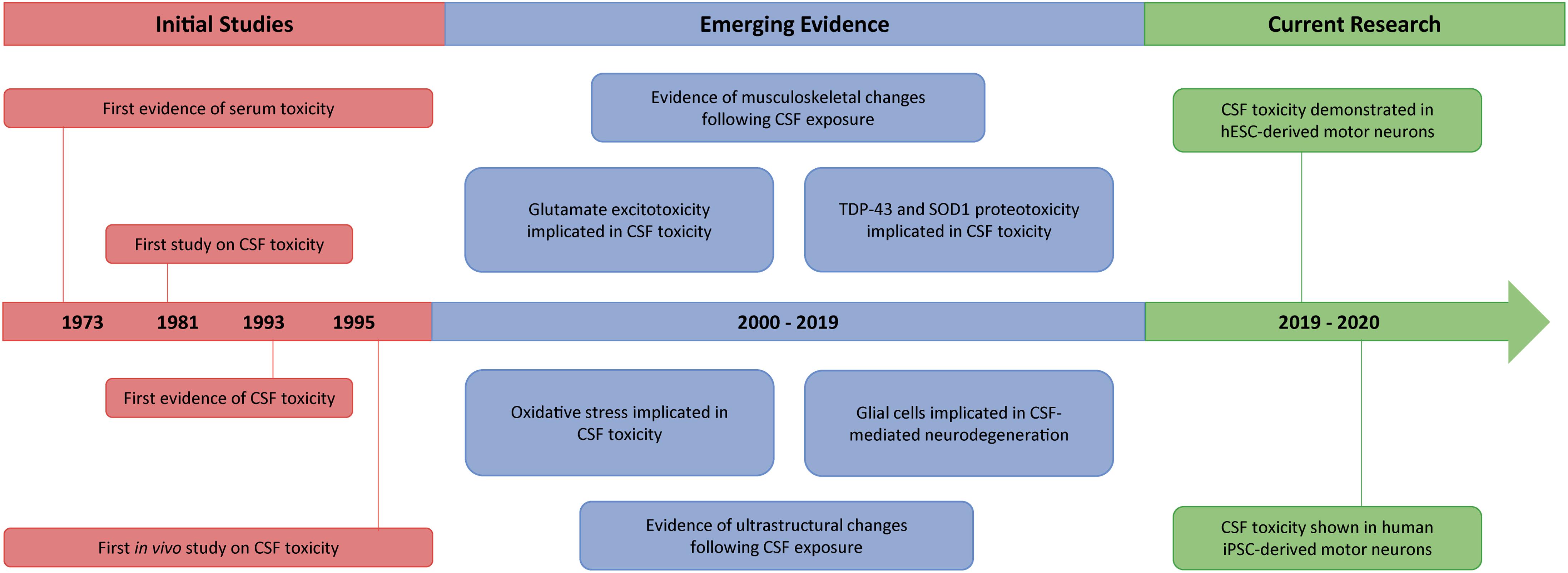
This prompted them to investigate the mechanisms that make these repeated dipeptides (R-DPRs) increasingly toxic. Kiskinis’ team hypothesized that this cascading effect is what leads to gain-of-function toxicity in motor neurons in this type of ALS. These repeats are transcribed and translated through non-canonical pathways which produce irregular RNAs and dipeptides (molecules with two amino acids bonded by a peptide) in neurons. In their paper published in Science Advances, the Kiskinis team studied a type of familial ALS caused by a repeat genetic sequence of the C9ORF72 gene, which is the largest genetic cause of ALS.Īccording to Kiskinis, healthy individuals may have 20 repeats of C9ORF72 in their genome, while patients with ALS can have thousands. There are two types of ALS: sporadic (non-genetic), which makes up more than 90 percent of all ALS cases, and familial (genetic). ‘Baiting’ Genetic MutationsĪn estimated 32,000 individuals are currently living with ALS in the U.S., according to the Les Turner ALS Foundation. The findings, published in Science Advances and Cell Reports, improve the understanding of ALS, a progressive neurodegenerative disease that attacks motor neurons in the brain and spinal cord, and provides support for the future development of targeted therapies. Evangelos Kiskinis, PhD, associate professor in the Ken and Ruth Davee Department of Neurology’s Division of Neuromuscular Disease and of Neuroscience, was senior author of studies published in Science Advances and Cell Reports.Ī pair of recent studies from the laboratory of Evangelos Kiskinis, PhD, associate professor in the Ken and Ruth Davee Department of Neurology’s Division of Neuromuscular Disease and of Neuroscience, have uncovered novel cellular mechanisms that are involved in two types of genetic amyotrophic lateral sclerosis, or ALS. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
